Cornea is an important tissue structure for refracting light and focusing imaging. Its transparency is a crucial factor to maintain vision. Corneal epithelial injury can be repaired through the proliferation of limbal stem cells. Corneal stromal injury will secondary to the disorder of extracellular stroma, secondary to corneal scarring, and corneal endothelial injury will cause corneal edema and opacity. Many primary corneal lesions, corneal trauma and corneal infection can significantly reduce vision and even lead to blindness. Corneal transplantation is an effective method to treat corneal scar and corneal endothelial decompensation, but the lack of corneal donors, rejection and graft survival limit the large-scale development of corneal transplantation. Therefore, seeking new corneal regeneration therapy is very important for corneal injury reconstruction and improving visual acuity. Corneal stromal stem cells, limbal stem cells, embryonic stem cells, adult stem cells, induced pluripotent stem cells and their secreted proteins and exosomes may participate in this process, which provides new opportunities and challenges for the clinical transformation of corneal regeneration. The treatment of corneal lesions may enter the era of cell therapy in the future. Recently, Professor Du Yiqin and his team summarized the relevant research on corneal regeneration treatment, and the relevant results were published in the journal progress in retinal and eye research (influencing factor 21.198).
Role of stem cells and their nutritional factors
Stem cells are cells that circulate slowly in the body and retain markers. They have the ability of self-renewal, clonal expansion and unlimited or long-term proliferation. Embryonic stem cells (ESC) and induced pluripotent stem cells (IPSC) can theoretically differentiate into all types of functional cells; Limbal stem cells (LSC), corneal stromal stem cells (CSSC), mesenchymal stem cells (MSc) and adipose derived stem cells (ASC) can differentiate into specific functional cells. Among them, LSC is located in the basal layer of limbal epithelium under Vogt grid, and CSSC is located in the specific stem cell fovea under the basal layer of LSC. The characteristics of corneal endothelial stem cells are not clear, which may be differentiated from dual stem cells located between corneal endothelium and trabecular meshwork. In transformation research and clinical practice, LSC has made great progress in the treatment of corneal chemical injury and thermal scald. There are also reports of exogenous CSSC preventing and reversing corneal scar.
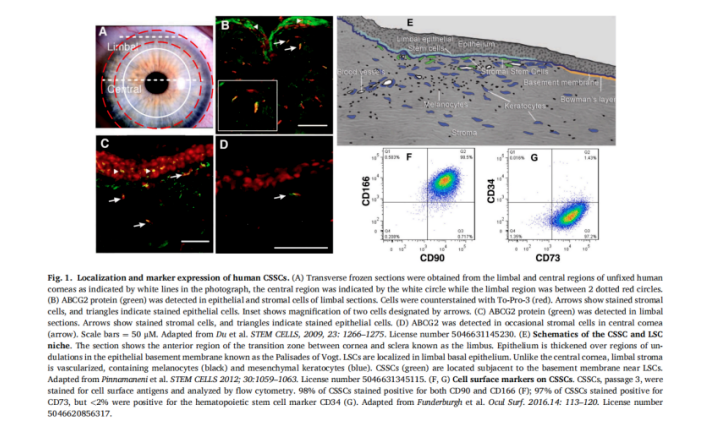
Exosomes, micro vesicles, extracellular vesicles and stem cell protein secretory group are the bridge between stem cells and the surrounding microenvironment. They contain soluble proteins, growth factors, cytokines, chemokines, miRNAs and bioactive lipids, which can widely regulate cell function. They have been reported in the treatment of retinal diseases and corneal injury healing. Because extracellular vesicles and exosomes can be extracted, separated and concentrated from cells to better play their functions, the cell-free therapy proposed by ratajczak et al. Has less damage to patients and better safety than stem cell therapy, which may be a new research field in the future.
Research progress of corneal regeneration induced by stem cells
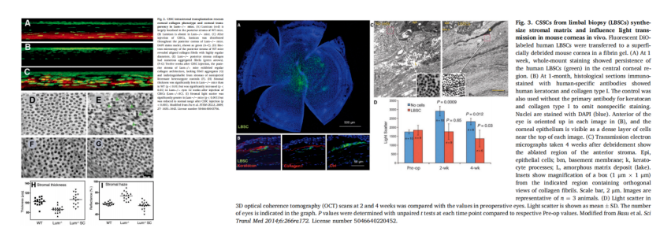
The distribution position of CSSC is relatively fixed, and its specific expression of ABCG2 and Pax 6 can be labeled by specific dyes Hoechst 33 and 342, which is convenient to isolate and retain its stem cell characteristics. CSSC was extracted from donors or patients by local biopsy as a source of corneal regeneration without affecting corneal function. Du et al. Had no obvious inflammatory reaction after injecting human CSSC into the corneal stroma of lum − / - mice lacking keratin sulfate polysaccharide. The diameter and spacing of the diseased corneal fibrous tissue were uniform. With the decline of corneal opacity, the corneal thickness and transparency returned to the normal level, and the CSSC still survived 4 months after transplantation. Basu was injected into the corneal surface to remove the elastic layer of corneal stroma, such as fibronectin C. After transplantation, CSSC secretes normal human keratin and type I collagen, and the regenerated corneal fibers are uniform and dense, which reduces the light scattering in the cornea and prevents scar formation.
In addition to human CSSC for corneal stromal regeneration, other stem cells from eyes and other tissues have also been used to study the potential of corneal regeneration. Embryonic stem cells (ESC) co cultured with mouse PA6 fibroblasts in vitro can induce the expression of cell markers such as CD271, p75NTR, sna11, ntrk3, Sox9 and Msx1. Under the remission of ascorbic acid, ESC can be induced to differentiate into keratinocytes and secrete keratin sulfate, which is a necessary molecule to maintain corneal transparency; Adipose derived stem cells (ASC) are derived from adipose tissue, which are easy to obtain and can effectively realize corneal regeneration. ASC was injected into the rabbit corneal stromal lysis model and successfully differentiated into functional corneal keratinocytes to repair corneal injury by expressing keratocan and ALDH3a1; Dental pulp stem cells (DPSC) have the same embryonic origin as corneal stromal cells and have the potential to differentiate into corneal stromal cells; Umbilical cord stem cells (umsc) can differentiate into corneal stromal cells after transplantation in the stroma, forming a wide range of dendrites, secretors and corneal proteins.
Application of corneal regeneration based on tissue engineering technology
The corneal stroma cultured in conventional medium in vitro is mostly lamellar structure, while three-dimensional culture on different types of nano biomaterial scaffolds can produce simulated natural corneal structure, which can be better used for in vivo transplantation to restore corneal transparency and improve vision. Composite ester polyurethane urea (PEUU) material is a scaffold structure with excellent biocompatibility. CSSC can differentiate into stromal cells after transplantation through fibroblast growth factor-2 (FGF-2) and transforming growth factor- β 3(TGF- β 3) Induction can deposit multiple layers of dense type I collagen fibers on the surface of the scaffold, and their regular arrangement can highly simulate human corneal stromal tissue. In addition, silk cellulose and polycarbonate can also be used as the scaffold structure of CSSC.
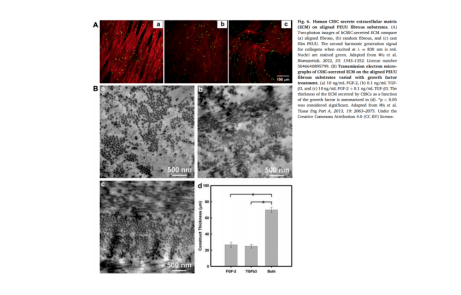
Although CSSC can produce corneal tissue in 3D three-dimensional nano biomaterial scaffolds, it is not easy to separate from the scaffolds, which limits the clinical application to a certain extent. Under the ideal 2D culture conditions of scaffold free tissue engineering, stem cells secrete a large amount of extracellular matrix to form tissue slices. Syed Picard et al. Cultured CSSC on the matrix with arranged micro grooves to form a layer of bionic corneal matrix tissue structure. In this structure, the collagen arrangement is consistent with the law, including type I and type V collagen and keratin, which is similar to natural human corneal matrix and can be easily removed by mechanical or chemical methods. Syed Picard et al. Implanted the regenerated corneal patch into the mouse stromal bag, which remained transparent and showed a curvature similar to that of the original cornea.
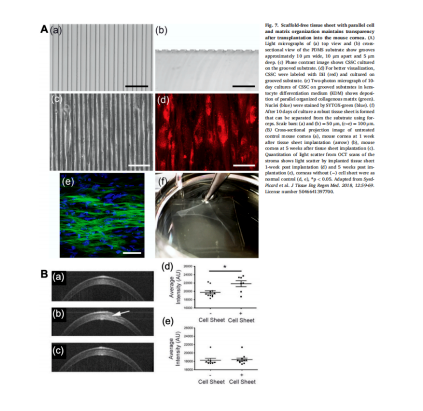
Prospect and Prospect of corneal regeneration therapy
At present, the curative effect of corneal regeneration therapy in different animal models is not stable. Formulating a unified quality control standard of stem cells before clinical transformation will greatly improve the success rate of treatment. In addition, assisted 3D bioprinting technology can design more innovative biological carrier materials, help to improve their compatibility and bionicity, reduce apoptosis and prevent complications such as inflammatory response and neovascularization. Topical application of exosomes and extracellular vesicles secreted by stem cells in the form of eye drops can accelerate the clinical transformation process and reduce the treatment damage. The action time of active ingredients on the corneal surface can be increased through the use of sustained-release gel, nano microspheres or bandage lens carrier. After corneal regeneration treatment, it opens up another new way for corneal blindness patients to see light again by further promoting corneal nerve regeneration and reversing corneal opacity.
References: Kumar a, Yun h, funderburgh ml, Du y. regenerative therapy for the cornea Progress in Retinal and eye Research. 2022 Mar; 87:101011. DOI: 10.1016/j.preteyeres. 2021.101011. PMID: 34530154.